Carbon-based refractories are a new type of refractory material developed in the late 1970s. By leveraging graphite’s high refractoriness and low wettability, they significantly enhance the high-temperature performance and erosion resistance of refractories. They exhibit excellent thermal shock stability, slag resistance, spalling resistance, and resistance to high-temperature creep. Aluminum-magnesium-carbon bricks are carbon-based refractories with an Al₂O₃ matrix. They are primarily composed of high-quality sintered high-alumina bauxite, with the addition of appropriate amounts of high-purity magnesia, flake graphite, and a binder, and are produced through heat treatment at temperatures of 200–300°C.
Magnesium-carbon bricks are unfired or lightly fired alkaline bricks manufactured using carbon-based materials and magnesia as raw materials, with a carbon-based binder. After being successfully developed in Japan, they were first used in hot spots of high- and ultra-high-power electric furnaces and in the gas nozzles of bottom-blown converters. Subsequently, Japan began applying magnesium-carbon bricks to various parts of converters. Compared to tar-impregnated and fired magnesia-dolomite bricks, the service life of converters is increased by approximately 1.3 to 1.6 times. While retaining the advantages of basic refractories, the introduction of carbon-based binders endows magnesium-carbon bricks with high refractoriness, strong slag resistance, low thermal expansion, and high thermal conductivity. This has significantly improved the performance of alkaline refractories, completely overcoming their traditional shortcomings, such as poor resistance to spalling and a tendency to absorb slag.
The following outlines the impact of the primary raw materials in aluminum-magnesium-carbon bricks and magnesium-carbon bricks on the final products:
Premium-grade sintered bauxite
The primary mineral components of high-alumina bauxite in China are gibbsite and kaolinite. Bauxite clinker is the product of high-temperature calcination; premium-grade bauxite clinker has an Al₂O₃ content of over 88.2%, reaching as high as 91.3%. Although Al₂O₃ exhibits strong resistance to erosion, pure Al₂O₃ has a high coefficient of thermal expansion and is susceptible to spalling. When used as a matrix, the Al₂O₃ matrix is easily penetrated and eroded by molten slag, exposing the aggregate and leading to structural spalling.
High-purity magnesia
Magnesia is produced by thoroughly sintering raw materials such as magnesite, brucite, and marine magnesium oxide at temperatures ranging from 1,600 to 1,900°C. Magnesia is classified into sintered magnesia and marine magnesia; sintered magnesia is produced from natural ores, while marine magnesia is produced from marine magnesium oxide. The main component of magnesia is magnesium oxide (MgO), along with small amounts of SiO₂, CaO, Fe₂O₃, B₂O₃, and other elements. It ranges in color from yellow to brown, with periclase as the primary crystalline phase. The grain size is 0.02–0.05 mm, and the density is 3.50–3.65 g/cm³. It exhibits excellent resistance to alkali slag erosion.
The key indicators for high-purity magnesia are MgO content, CaO/SiO₂ ratio, microstructure, and bulk density. Magnesia with high MgO content has periclase as its primary crystalline phase and contains few impurity cementation products; refractory materials produced from it exhibit extremely high resistance to melt erosion. The CaO/SiO₂ ratio determines the phase composition of the matrix in magnesia, directly affecting the bonding of periclase and the high-temperature performance of the refractory material. Generally, magnesia with a C/S ratio of 3–8 exhibits better high-temperature performance; values outside this range may have adverse effects; Microstructure is a key method for characterizing the grain size and bonding state of periclase. Typically, grain sizes of 80–150 μm are required. Bulk density is an important indicator of the sintering degree and compactness of magnesia. Magnesia with a higher bulk density can resist slag intrusion and enhance the corrosion resistance of refractory products.
Electrofused magnesia
Electrofused magnesia, also known as electrofused magnesium oxide, is produced by melting magnesite or sintered magnesia in an electric arc furnace at a high temperature of 2500°C, followed by cooling and crushing. The purity of electrofused magnesium oxide is determined by the purity of the raw materials. Its primary crystalline phase is periclase; the periclase crystals that crystallize from the melt are coarse and densely structured, with a high degree of crystal-to-crystal contact. It exhibits excellent resistance to water and slag in atmospheric conditions, as well as good high-temperature volume and chemical stability, remaining stable in an oxidizing atmosphere at 2300°C. The performance indicators of magnesia-carbon bricks are directly related to those of magnesia sand. To improve the performance of magnesia-carbon bricks, it is necessary to increase the MgO content, thereby enhancing the direct bonding of periclase within the magnesia sand. Controlling the C/S ratio can reduce the amount of silicates and minimize the degree to which periclase is fragmented by silicate phases. Therefore, magnesium content and the calcium-to-silicon ratio are key indicators for evaluating magnesia, as shown in Table 1.
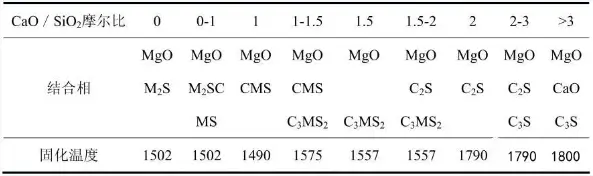
As shown in Table 1, the optimal calcium-to-silicon ratio is less than 1 or greater than 1; a high calcium-to-silicon ratio helps improve the stability of magnesium coexisting with graphite at high temperatures. Both the size of the periclase grains and the bulk density of the magnesia sand have a significant impact on the erosion resistance of magnesia-carbon bricks. Akira Matsuo and colleagues studied the fabrication of magnesia-carbon bricks using magnesia with magnesite grains of different diameters and measured their weight loss under high-temperature reducing atmospheres. The results indicated that for magnesia-carbon bricks made from magnesia with magnesite grains of different diameters, the larger the magnesite grain size, the smaller the weight loss. It can thus be concluded that, when producing high-performance magnesia-carbon bricks, one should select electrofused magnesia with a CaO/SiO ratio of <1 or >2, high bulk density, and well-developed crystal morphology as the raw material.
Graphite
Graphite possesses excellent thermal conductivity and refractory properties, with a melting point as high as 3,500°C. It has a low coefficient of thermal expansion (1.4×10⁻⁶ at 1,000°C), high thermal conductivity, and good resistance to rapid cooling and heating; it is one of the few materials whose strength increases with rising temperature. Graphite also has a relatively large contact angle with slag and does not form eutectic mixtures with Al₂O₃, SiC, or SiO₂, thereby preventing slag from penetrating the product. Since carbon can reduce iron oxide in the molten slag to metallic iron, it increases the viscosity of the slag, reducing the migration of slag components into the brick and achieving an erosion-reducing effect.
The primary function of graphite in carbon-containing products is to effectively prevent slag from penetrating the brick structure. It achieves this by increasing the contact angle between the brick’s working surface and the slag, and by reacting with the MgO in the brick to reduce magnesium to metallic form while producing CO gas. The pressure of this gas prevents slag penetration, while magnesium diffuses, volatilizes, and oxidizes on the brick’s working surface, forming a dense, impermeable MgO layer. This creates a strongly reducing environment within the brick, which not only reduces iron oxides in the slag but also increases slag viscosity, thereby preventing slag from penetrating the brick. Graphite with higher carbon content and larger flakes exhibits greater resistance to erosion and slag corrosion. The SiO₂ content in graphite has the following relationship with the erosion index of magnesium-carbon bricks, as shown in Figure 1:
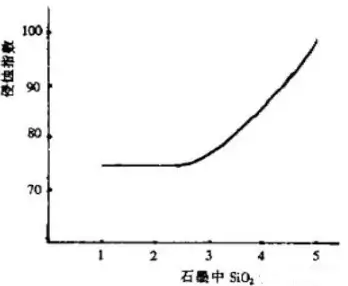
As shown in Figure 1, the erosion index increases continuously with rising SiO₂ content, while erosion resistance decreases steadily. When the SiO₂ content in the graphite exceeds 3%, the erosion index of the magnesium-carbon brick rises sharply, and its erosion resistance drops dramatically. As the flake size of the graphite in the magnesium-carbon brick increases, their oxidation resistance increases. However, when the flake graphite particle size exceeds 0.125 mm, the rate of increase in oxidation resistance slows down, making 0.125 mm the optimal particle size. Since graphite readily oxidizes to form CO, oxidized graphite loses these excellent properties, thereby reducing the refractory material’s erosion resistance. This is a critical weakness of graphite and a major cause of damage to carbon-containing materials.
Binder
Although the binder content in the final product is not high, it is one of the key technical factors in the production of carbon-containing products. The binder directly affects the mixing and forming properties of the green body, as well as the microstructure of the final product. During mixing and forming, the binder must exhibit good wetting properties toward refractory aggregates and graphite, and possess an appropriate viscosity to improve the quality of the green body mix and the bulk density of the green body.
The main characteristics of the binder are:
(1) Good wettability: It exhibits good wettability toward both magnesia and graphite;
(2) It contains no or minimal components harmful to humans;
(3) The properties of the mixed slurry remain relatively stable over time, and it causes minimal chemical reactions with the aggregates;
(4) During the heating process of the product, the binder should maintain a high residual carbon content, and the carbonized polymer should possess good high-temperature strength.
Only with good wettability can the binder be distributed relatively uniformly on the surfaces of the particles and graphite, forming a continuous network structure as much as possible. Only then can a continuous carbon skeleton be formed after carbonization, thereby improving the strength and erosion resistance of the product. The type of binder and carbonization conditions directly affect the microstructure and properties of the bound carbon; differences in the carbonization process of the binder also result in significant variations in the structure of the resulting bound carbon. To ensure that the formed brick blanks possess sufficient strength, thermosetting phenolic resins are typically used as binders in the production of aluminum-magnesium-carbon bricks and magnesium-carbon bricks. The resin used should have appropriate viscosity, a high carbon content, and a high residual carbon rate. Phenolic resin, a substitute for coal tar pitch rich in benzo[a]pyrene, is produced by the reaction of phenol and formaldehyde. Depending on the reaction conditions, the product is either phenolic varnish resin or A-class phenolic resin. Because phenolic resins do not exhibit thermoplastic behavior when heated, the dimensional accuracy of the final product can be ensured. Compared to graphite, the carbonized products of the resin form a banded crystal lattice that stacks to create a layered structure (polycarbon or vitreous carbon). During high-temperature decomposition, phenolic resin first releases water (from the A-phase phenolic resin), phenol, cresol, and small amounts of xylenol and formaldehyde, ultimately forming polymerized carbon.
Synthetic resins can be thoroughly mixed with refractory particles at room temperature without the need for heating; their residual carbon content is similar to that of asphalt, ranging from 50% to 70%. The main drawbacks of liquid A-class phenolic resins are: limited stability; the homogeneity of the resin’s carbonization products makes them prone to oxidation, reducing their resistance to erosion; and they are relatively sensitive to thermomechanical stress. The addition of rapidly oxidizable metal additives, such as Al, Mg, and Si, to the mixture is intended to compensate for these shortcomings.
Additives
It is precisely the presence of graphite that gives carbon-based refractory materials their excellent slag resistance and thermal shock stability; damage to these materials is primarily caused by the oxidation of graphite. Once graphite is oxidized, its advantages are virtually lost. To improve the oxidation resistance of carbon-based refractory materials, small amounts of additives such as Si, Al, Mg, Zr, SiC, and BC are often incorporated.
The following analysis examines the mechanism of action of these additives from both thermodynamic and kinetic perspectives:
From a thermodynamic perspective, the mechanism of action is as follows: at operating temperatures, the affinity of the additives—or the products formed by their reaction with carbon—for oxygen is greater than that of carbon for oxygen. Consequently, these substances are oxidized before the carbon, thereby protecting it.
From a kinetic perspective, the compounds formed by the reaction of additives with O₂, CO, or carbon alter the microstructure of carbon-based refractory materials. This includes increasing density, blocking pores, and hindering the diffusion of oxygen and reaction products.
Non-oxide additives in carbon-based refractory materials typically serve the following functions:
(1) Reducing carbon monoxide to carbon to suppress the rate of carbon consumption;
(2) Forming carbides or oxides to enhance the degree of densification of the refractory material;
(3) Further promoting graphite crystallization;
(4) Reducing open porosity;
(5) Forming a protective layer;
(6) Improving high-temperature strength.

