Currently, the refractory material primarily used for the working lining of continuous casting ladles is magnesium-based dry mix. This dry mix offers excellent resistance to erosion by high-iron and alkaline slag, a long service life, no contamination of molten steel, ease of installation, and ease of ladle removal and turning. It has gradually been adopted for ladle working linings and has demonstrated good performance and promising prospects. However, magnesium-based dry mixes generally have a high density, and this type of dry mix has the following disadvantages:
1) Due to their high thermal conductivity, heat dissipates rapidly from the ladle lining, causing the molten steel to cool quickly and consuming a significant amount of thermal energy.
2) Because of their high density, the refractory consumption per ton of steel in the ladle remains high, wasting mineral resources and increasing the company’s refractory costs.
3) Magnesium oxide has a high coefficient of thermal expansion, poor thermal shock resistance, and is prone to moisture absorption and hydration. Additionally, it exhibits poor resistance to thermal spalling and structural spalling, which significantly reduces the service life of the refractory material.
In addition, the dry lining material currently used in ladles is primarily bonded with phenolic resin. During the baking process, as the ladle temperature rises, the phenolic resin gradually cures to form a carbon network structure, giving the dry lining material good strength. However, dry lining materials using phenolic resin as a binder introduce carbon enrichment into the molten steel, which significantly affects the quality of clean steel production. At the same time, the cured resin decomposes at temperatures between 200°C and 800°C, releasing gases such as CO₂, CO, CH₄, H₂, and H₂O. The combination of free phenol and these generated gases forms flue gas with a pungent odor, posing a certain degree of health hazard to on-site construction personnel. Therefore, existing refractory materials used for ladle working linings no longer meet the requirements for cleanliness and thermal insulation performance.
This paper focuses on the research and development of a resource-saving, lightweight, and environmentally friendly dry mix for ladles. With low natural bulk density and volume density, this material not only reduces refractory consumption per ton of steel and improves the thermal insulation performance of the dry mix but also employs eco-friendly binders that do not produce harmful or irritating gases, making it highly environmentally friendly.
Test
1.1 Materials and Experimental Protocol
Using sintered magnesia, magnesite, and olivine as the main raw materials (see Table 1 for physical and chemical properties), the mixture is formulated in specific proportions using particles of 5–1 mm, ≤1 mm, and 200 mesh, with the addition of binders and additives, supplemented by a small amount of paper fibers.
1.2 Test Procedure and Performance Testing
Prepare the ingredients according to the designed formula, using a specialized steel cup as the container, and weigh its mass as m1. Place each batch of thoroughly mixed material onto the bulk density tester, position the steel cup at the bottom, fill it with the material until full, and level the surface so that the sample naturally fills the cup. Weigh the sample’s mass as m2. Calculate the sample mass m = m2 – m1, and calculate the bulk density as mass m / volume V.
Place each batch of thoroughly mixed material into a mixer, mix evenly, then pour it into a 160 mm × 40 mm × 40 mm triple-cavity cement specimen mold and tamp it by hand to form the specimen. When tamping the rectangular specimen, first spread a layer flat and then compact it, repeating until the mold is full. Place the compacted specimens, along with the molds, into an oven and bake at 200°C for 3 hours. Remove from the molds after cooling.
Determine the bulk density, compressive strength at room temperature, and linear change rate after heat treatment at 200°C for 3 hours and 1500°C for 3 hours, respectively, to evaluate the specimens’ low-temperature bonding performance and sintering performance. Additionally, pour the mixed dry mixture into a circular mold with a diameter of 180 mm and a thickness of 20 mm, compact it by hand, place it in an oven, and hold it at 200°C for 3 hours before demolding. Measure its thermal conductivity at different temperatures.
2.1 Bulk Density and Volume Density
Figure 1 shows the bulk density and volumetric density of the dry material.
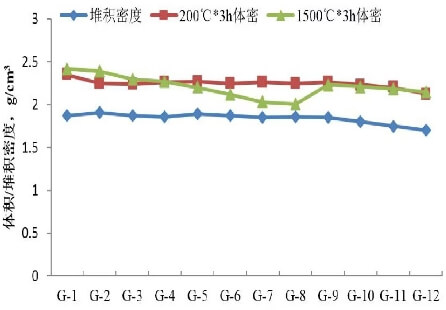
As shown in Figure 1, replacing phenolic resin powder with glucose resulted in little change in the bulk density of the dry mixture; however, the volume density decreased slightly after holding at 200°C for 3 hours. When 6% (w) glucose was added, the volume density decreased by approximately 5%. However, with the addition of magnesite and olivine, both the bulk density of the dry mixture and the volume density after holding at 200°C for 3 hours showed little change. With the introduction of paper fibers, the bulk density of the dry mixture and the volume density after holding at 200°C for 3 hours decreased significantly. When 0.1%, 0.2%, and 0.3% (w) of paper fibers were added, the bulk density decreased by 2.7%, 5.4%, and 8.1%, respectively, and the volume density decreased by 1.3%, 2.6%, and 5.7%, respectively. Comparing samples G1 and G12, with glucose used as a binder and 40% (w) olivine and 0.3% (w) paper fibers added, the bulk density and volume density of sample G12 both decreased by approximately 9%. This reduction in bulk and volume density enables material lightweighting, reduces the consumption of refractory materials per ton of steel in ladles, and lowers the company’s refractory costs. Figure 1 also shows that as the amount of magnesite added increases, the bulk density after a 3-hour soak at 1500°C exhibits a decreasing trend.
Figure 1 shows that the introduction of magnesite and olivine, which have lower bulk densities, had little effect on the bulk density of the dry mixture or its bulk density after being held at 200°C for 3 hours. It is estimated that the bulk density of magnesite and olivine is approximately 7.9% lower than that of calcined magnesia. Based on an addition ratio of 40%, this could theoretically reduce the bulk density by about 3.1%. However, many factors influence bulk density, such as the particle shape of the raw materials. Particle shape is related to the structural characteristics of the raw materials and the crushing method, resulting in variations in both particle shape and size. Therefore, this study did not clearly demonstrate their impact on the bulk density of the dry mixture, and further investigation is required.
The use of glucose as a binder has a significant effect on reducing the bulk density and volume density of dry materials. This is believed to be primarily due to differences in particle size: glucose particles are coarser than phenolic resin powder, while the finer particle size of the phenolic resin powder allows for more uniform filling of the pores. The figure also shows that the bulk density after holding at 1500°C for 3 hours follows a similar trend to that after holding at 200°C for 3 hours. However, for samples G-5 to G-8 containing magnesite, the bulk density after holding at 1500°C for 3 hours decreased significantly and declined linearly with increasing magnesite content. This is because magnesite begins to decompose at approximately 400°C during the baking process, releasing CO₂ gas. The reaction intensifies when the temperature reaches 550–650°C, and by 1000°C, decomposition is complete, producing calcined MgO. This material is porous, has a high porosity, and a low bulk density, thereby reducing the bulk density of the dry mixture after holding at 1500°C for 3 hours.
2.2 Physical Properties at Room Temperature
Figure 2 illustrates the effect of different raw materials on the physical properties of dry mixtures. As shown in the figure, compared to phenolic resin, the compressive strength at room temperature of specimens using glucose as a binder decreases after being held at 200°C for 3 hours and at 1500°C for 3 hours. As the amount of glucose added increases, the compressive strength of the specimens gradually increases. at a glucose content of 6% (w), although the compressive strength of the bonded samples still cannot match that of the phenolic resin-bonded samples, it is fully capable of meeting the on-site demolding requirements of customers. Phenolic resin forms a relatively robust three-dimensional network structure after low-temperature heat treatment. Glucose has a melting point of 146°C; during baking at 200°C, it undergoes cross-linking and condensation reactions to form a three-dimensional network structure. Furthermore, water and other byproducts generated by the decomposition of glucose during heating can react chemically with magnesium oxide, thereby imparting strength to the dry mixture. Glucose is non-toxic and harmless; it does not produce irritating gases such as phenol or formaldehyde during baking. It offers advantages such as being environmentally friendly and cost-effective, making it an eco-friendly binder to replace phenolic resin. As shown in Figure 2, when the glucose content is further increased to 7%, the compressive strength of the dry mixture at room temperature does not increase significantly. However, the linear change rate after heat treatment at 1500°C for 3 hours is substantial. Therefore, a recommended addition ratio of 6% (w) is suggested.
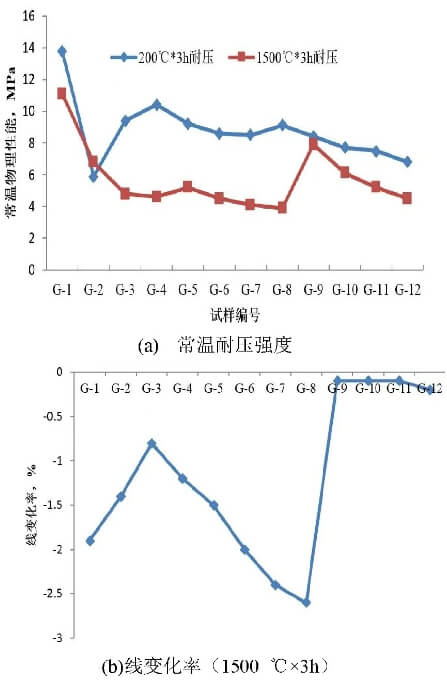
As the proportion of magnesite increased, the compressive strength of the specimens at room temperature remained largely unchanged after holding at 200°C for 3 hours; however, after heat treatment at 1500°C for 3 hours, the compressive strength showed a gradual decline, and the linear change rate exhibited the same trend. This was primarily due to the decomposition of magnesite during the baking process, which released gases, resulting in a loose texture and high porosity.
As the proportion of paper fibers increases, the compressive strength of the specimens decreases after both 3-hour heat treatments at 200°C and 1500°C. Analysis suggests that the introduction of paper fibers hinders the material’s ability to pack tightly, impairs the effectiveness of the binder, and creates internal pores upon volatilization during baking, thereby reducing physical properties. When 0.3% paper fiber is added, the material’s physical properties at room temperature still meet on-site demolding requirements. Additionally, as paper fibers shrink and burn during baking, they form microscopic pores and channels within the dry mixture. This facilitates the rapid escape of gases generated by binder decomposition during baking, alleviates internal stress, and reduces the risk of working liner collapse.
As shown in Figure 1(b), the linear change rate after heat treatment at 1500°C for 3 hours gradually increases with the addition of magnesite. This is attributed to the decomposition of magnesite during the drying process, which releases gases and results in a porous structure; this, in turn, leads to sintering shrinkage during high-temperature heat treatment. For specimens containing olivine, the linear change rate after heat treatment at 1500°C for 3 hours was significantly reduced. Analysis suggests that this may be due to further reaction between MgO and SiO₂ to form the olivine phase; the slight expansion of this phase fills the pores, thereby reducing the linear change rate. Additionally, this improves the material’s resistance to slag penetration.
2.3 Thermal Conductivity
Figure 3 shows a comparison of the thermal conductivities of specimens G-1, G-3, G-9, and G-12 at different temperatures. It can be seen that the thermal conductivity decreases at all temperatures. The thermal conductivity of G-3 shows a significant decrease compared to G-1 at 600°C, 800°C, and 1000°C. This is attributed to the fact that phenolic resin forms a carbon network structure upon carbonization, thereby enhancing thermal conductivity, whereas G-3 uses glucose as a binder, resulting in a significant reduction in residual carbon and, consequently, a decrease in thermal conductivity. Compared to Sample 3, the thermal conductivity of Sample G-9 is further reduced. This is attributed to the fact that the thermal conductivity of olive sand is lower than that of magnesia sand (6.7 W·m⁻¹·K⁻¹ at 1000°C). In sample G-12, the introduction of paper fibers reduced the bulk density and created microscopic pores during the baking process, increasing the porosity. This reduced the formation of low-melting substances and effectively prevented them from aggregating, making sintering difficult and lowering the thermal conductivity.
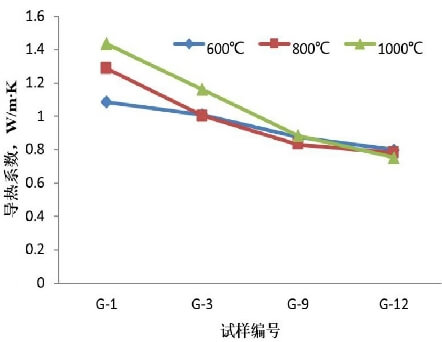
A lower thermal conductivity, on the one hand, helps create a larger temperature gradient across the working layer from the hot side to the cold side. This facilitates gradual sintering of the working layer into the permanent layer, resulting in a dense structure on the hot side of the working layer and preventing phenomena such as through cracks that could allow slag to penetrate into the permanent layer. On the other hand, it slows down the heat dissipation rate of the ladle lining, thereby improving its thermal insulation properties and reducing the shell temperature. As a result, the molten steel cools more slowly during operation, leading to significant energy savings.
(1) By replacing phenolic resin with non-toxic, harmless glucose as a binder, the material achieves good demolding strength while producing no irritating gases such as phenol or formaldehyde during the baking process. This approach offers advantages such as environmental friendliness and low cost.
(2) The incorporation of paper fibers reduces the bulk density, volume density, and thermal conductivity of the dry mix, achieving material lightweighting and contributing to energy conservation and reduced consumption.
(3) Using glucose as a binder and incorporating magnesium olivine and paper fibers both help reduce thermal conductivity, slowing the heat dissipation rate within the ladle lining and thereby enhancing its thermal insulation performance.
